Effects of a low carbohydrate diet on energy expenditure during weight loss maintenance: randomized trial (Ebbeling et al., 2018)
This is another NuSi study designed to evaluate certain aspects of the carb/insulin model of obesity. Understanding the design of this study is critical to being able to properly evaluate the results.
Very important:
1. All food was provided, all the diets were healthy, and most people complied.
2. This was a very expensive study. They used great methods.
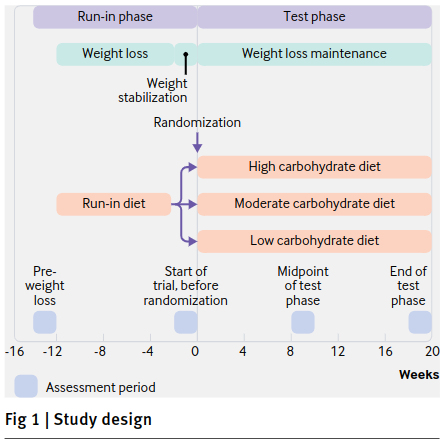
Study design: during the “Run-in diet,” everyone followed the same diet (C/F/P: 45%/30%/25%) at 40% caloric restriction in order to lose about 10% of their initial body weight.
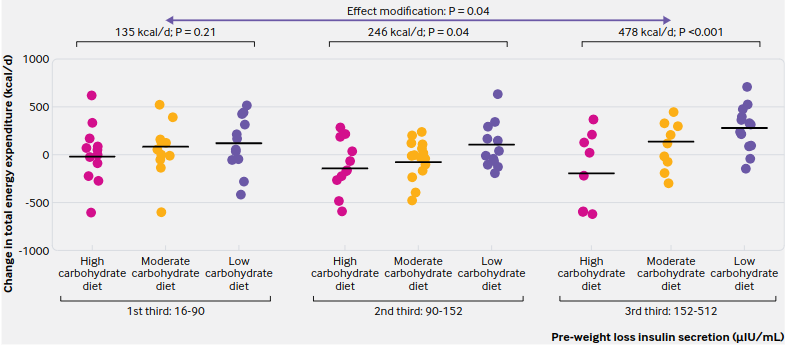
Importantly, insulin sensitivity was assessed and this may have influenced what happened next, in the weight maintenance phase. I used to put a lot of weight on this theory — eg, the top 25% most insulin sensitive people will do better on low fat whereas the bottom 25% insulin sensitive people (the most insulin resistant, ie) will do better on low carb — this theory has fared better or worse depending on which study you look at. In this study, it did pretty well.
In the end, we had 38 people in the entering into the high carb arm of the weight maintenance phase and 43 on low carb.
Remember, they all lost weight on the same diet. Now they’re being fed enough to maintain body weight (low carb, moderate carb, or high carb) and we’re measuring things.
Really exciting stuff!
For the rest of this article and more, head over to Patreon! Five bucks a month for full access and there are many other options. It’s ad-free and you can cancel if it sucks 🙂
If you’re interested in setting up consultations with me, reach out: drlagakos@gmail.com.
Affiliate links: Still looking for a pair of hot blue blockers? TrueDark is offering 10% off HERE and Spectra479 is offering 15% off HERE. If you have no idea what I’m talking about, read this then this.
Join Binance and get some cryptoassets or download Honeyminer and get some Bitcoins for free!
20% off some delish stocks and broths from Kettle and Fire HERE.
If you want the benefits of ‘shrooms but don’t like eating them, Real Mushrooms makes great extracts. 10% off with coupon code LAGAKOS. I recommend Lion’s Mane for the brain and Reishi for everything else.
Join Earn.com with this link.
Start your OWN Patreon campaign!