Conventional leptin resistance has something do with obesity. It is known. Silent leptin resistance is … err … complicated.
Divide and conquer
Fructose-induced leptin resistance exacerbates weight gain in response to subsequent high-fat feeding (Shapiro, Scarpace, et al., 2008 AJP)
A remarkable 60% fructose diet fed to rats for 6 months had absolutely no effect on energy balance. Nil. QED.
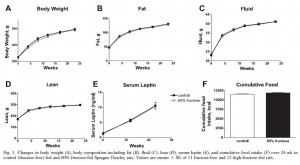
Food intake and body weight were unaffected because the levels of and sensitivity to endogenous leptin were identical in both groups.
Enter the Dragon
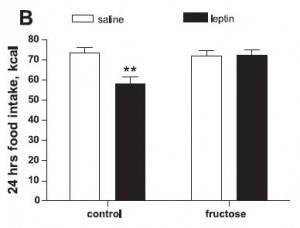
“Silent Leptin Resistance” – The fructose-fed rats are, however, profoundly resistant to the satiating effects of Metreleptin (a pharmaceutical grade injectable leptin analog):
Lustig says Leptin Resistance is obesity. These data say that Silent Leptin Resistance could predispose to obesity. The figure above shows resistance to the satiating effect of Metreleptin in fructose-fed rats. Thus, Silent Leptin Resistance can be accurately gauged by measuring food intake in response to Metreleptin. Recall the levels of and sensitivity to endogenous leptin is identical in these two groups (compare the two open bars in this graph or E & F in the figure above). Silent Leptin Resistance is unmasked only after pharmacological intervention. But since the rats had normal body weight and food intake, what is the relevance of Silent Leptin Resistance? These rats were markedly predisposed to obesity.
As I’ve blogged about previously (the great difficulties of weight loss), the correlations of leptin with 1) body weight, and 2) specific measurements of appetite like “hunger” and “urge to eat” are not very strong. Further, not only does leptin fail to accurately gauge appetite, neither leptin levels nor appetite correlate very well with weight re-gain in humans (Sumithran et al., 2011). In this study, it appears as though this confusion is specific to endogenous leptin, as the effects of Metreleptin seem to accurately quantify some meaningful aspect of Silent Leptin Resistance and predict the propensity for weight gain (see below).
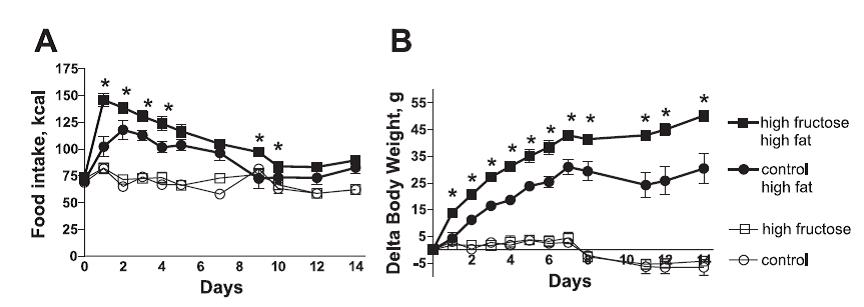
The most interesting data have already been presented, but the editorial money bag is the next figure. Half the rats from each group were switched to moderate sugar high fat diets and the fructose pre-fed rats became significantly more obese than the low sugar fed controls.
Both groups switched to high fat ingested more calories initially, but those pre-fed high fructose diets gained significantly more weight than those pre-fed the low fructose diet. Thus, while the physiology of endogenous leptin was proper in its inability to detect differences in the fructose-fed rats PRIOR to the diet switch, when the rats were for all intents and purposes equally leptin sensitive, it was only the response to Metreleptin that predicted the propensity for weight gain after exposure to an obesogenic diet.
Fructose did not dysregulate endogenous leptin sensitivity, and accordingly, food intake, body weight, and leptin levels were similar in fructose-fed and control rats prior to the diet switch. But fructose loaded the gun, Metreleptin revealed a bullet in the chamber, and the obesogenic diet pulled the trigger. What do you think is going on in those [rare] skinny people with Lean Metabolisms who eat HFCS-rich foods and chug soda all day long?
I don’t know the full implications of what would be revealed by further investigations into Silent Leptin Resistance, but I know that none of this bodes well for sugar.