From the best I can gather, one of the more immediate players in circadian biology is the coenzyme nicotinamide adenine dinucleotide (NAD), which participates in a variety of redox reactions. Fasting increases the intracellular NAD/NADH ratio, setting off a cascade of events involving epigenetics and the regulation of metabolism.
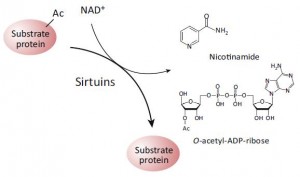
NAD activates sirtuins, a family of deacetylase enzymes. This is epigenetics.
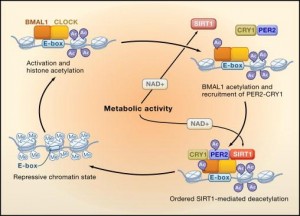
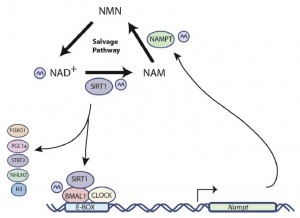
SIRT1 regulates the activity of BMAL1 and CLOCK, two circadian transcription factors, which target NAMPT, an enzyme that synthesizes NAD. And in a curious feed-forward mechanism, CLOCK and BMAL1 enhance SIRT1 expression… genetic deletion of any of these players induces insulin resistance (Zhou et al., 2014), and this can be recapitulated with constant darkness: reduced BMAL1 and SIRT1, hepatic insulin resistance; the latter can be reversed with resveratrol (which may or may not be acting through SIRT1; this is controversial). While alcohol does no great favors for circadian biology, if you’re going to imbibe, perhaps a resveratrol-rich Argentinian malbec served, and this might be the important part, at night, when all of this stuff is going on… coincidentally [fortunately], that’s precisely when most choose to imbibe.
The downside of drinking alcohol [at night] is that ethanol metabolism consumes NAD, potentially throwing a wrench into the gears of this particular aspect. Perhaps supplementing the NAD precursor, nicotinamide riboside, would be helpful? “It has properties that are insulin sensitizing, enhancing to exercise, resisting to negative effects of high-fat diet, and neuroprotecting” (Chi et al., 2013)…
fasting -> NAD -> SIRT1 -> BMAL1/CLOCK -> NAMPT -> NAD
Viewed this way, fasting could potentially be seen as a circadian reset biohack. And in a sense, we already do this [when we’re supposed to]: while sleeping [at night]. SIRT1 also activates PGC1a in liver (Rehan et al., 2014), which enhances fatty acid oxidation, at a time when we need it: while sleeping, which [fortunately] occurs at night.
All of these coincidences… you might begin to think we’ve been formulated throughout all of evolution with daily light-dark cycles that fundamentally dictated how we now operate.
Some people supplement with the amino acid tryptophan at night to help them sleep. Tryptophan is a precursor of melatonin
, a sleep-inducing, energy-conserving hormone. Tryptophan is also a precursor of NAD, and people take it at night, when the redox potential is expected to be biased toward NAD, because we’re fasting, because we’re sleeping…
This is epigenetics.
Part 2. Animals with disrupted circadian genes in just about any tissue or cell-type exhibit deranged metabolisms in one way or another (eg, muscle: Dyar et al., 2013; liver: Zhou et al., 2014; macrophages: Xu et al., 2014)… and given the circadian nature of the redox potential, and the observation that insulin sensitivity also shows a bona fide diurnal variation (eg, Shi et al., 2013, blogged about here), it’s really no surprise that restricted feeding windows can completely reverse diet-induced obesity and insulin resistance in animal models, and in some cases this is independent from other factors like food intake and physical activity (eg, Hatori et al., 2011; Sherman et al., 2012; Shamsi et al., 2014).
Mind your rhythms.
If you like what I do and want to support it, check out my Patreon campaign!
UPDATED Affiliate Links: It’s 2018, join Binance and get some damncryptoassets or download Honeyminer and get some Bitcoins for free!
Start your OWN Patreon campaign!
Join Earn.com with this link and get paid to answer questions online.
Still looking for a pair of hot blue blockers? Carbonshade is offering 15% off with the coupon code LAGAKOS and Spectra479 is offering 15% off HERE.
If you have no idea what I’m talking about, read this then this.
20% off some delish stocks and broths from Kettle and Fire HERE.
If you want the benefits of ‘shrooms but don’t like eating them, Real Mushrooms makes great extracts. 10% off with coupon code LAGAKOS. I recommend Lion’s Mane for the brain and Reishi for everything else.