Open access for all at Patreon! <- linkage
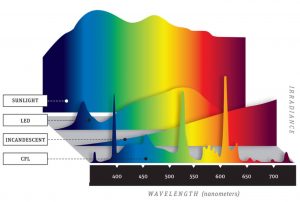
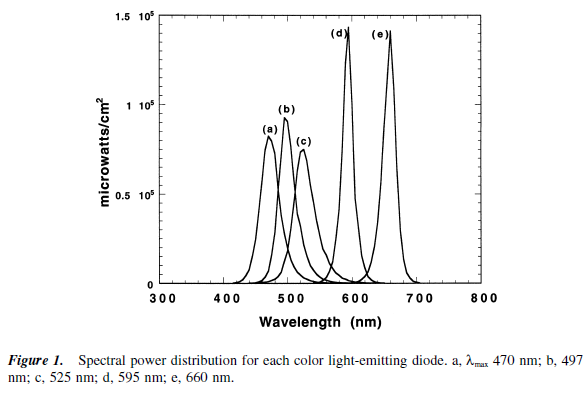
Approximate wavelengths, in nanometers (nm):
680 red
595 amber
525 green
497 blue/green
470 blue
[Strongly] suggested pre-reading: Artificial light and circadian rhythms: blocking the blues and The Hot Blue-Blocker Experiment
Patrons can just click here…
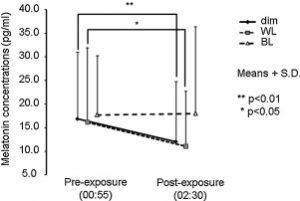
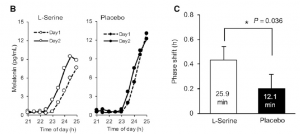
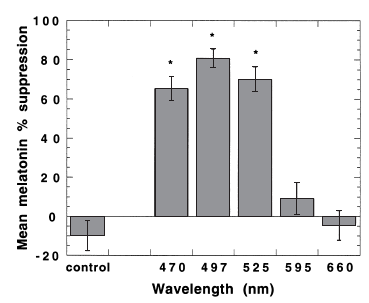
Study 1 tested the effects of different wavelengths of light exposure (compared to total darkness) from midnight til 2 AM on melatonin suppression on night 1 and melatonin onset on night 2 (Wright and Lack, 2001). Note: there was no light on night 2.
On night 1, they found that 470 (blue), 497 (blue/green), and even 525 (green) suppressed melatonin, ranging from 65 to 81%.
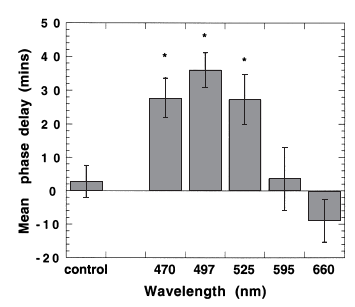
However, remarkably, on night 2 those same wavelengths had a carry-over effect, delaying melatonin onset by 27 to 36 minutes!
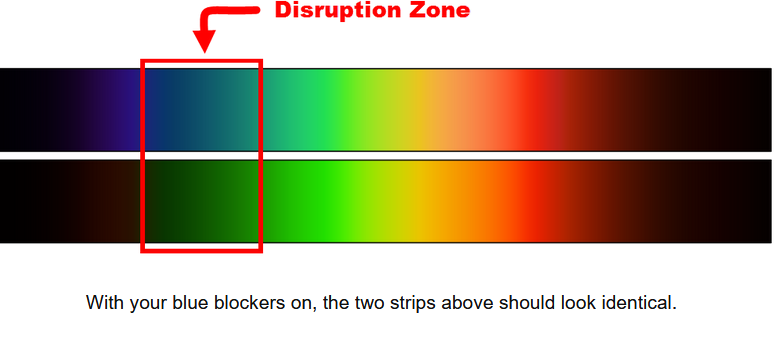
This is why lens color of your blue blockers matters. Orange lenses block blue, although blue/green and even green can still have a detrimental impact. Redder lenses more effectively block in the green range.
If you get up to pee or whatever in the middle of the night, it might be prudent to rock your blue-blockers and/or have a lamp with a red bulb.
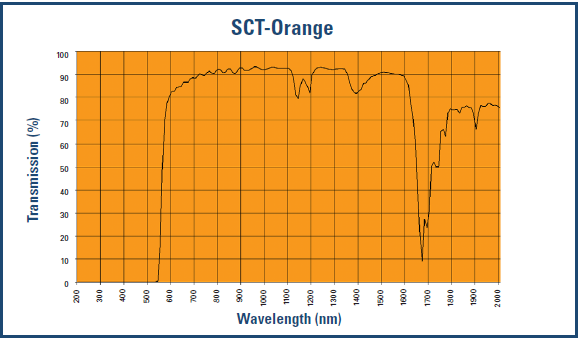
The following graphs show you how much light is blocked by different lenses – remember, we want as little transmission up to around 525 nm (according to study 1 [above] and study 2 [below]).
However, for a quick and dirty test you can do at home, the people at Spectra479 put this together:
Carbonshade and Spectra479 are offering 15% off if you enter the coupon code LAGAKOS at checkout.
Normal gray-lensed Ray-Bans block about 85% of all light. Cool for blocking UV, but you’re still getting about ~15% of blue and green light. That’s too much.

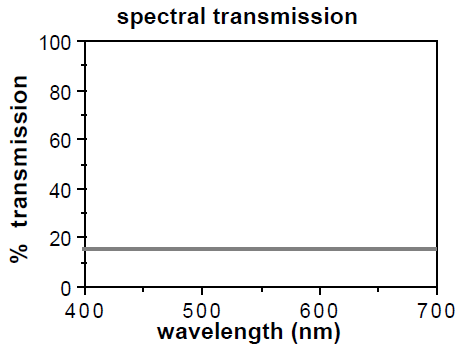
Spectra479s block 99.8% of 450-510nm, which fully encompasses blue to blue-green.

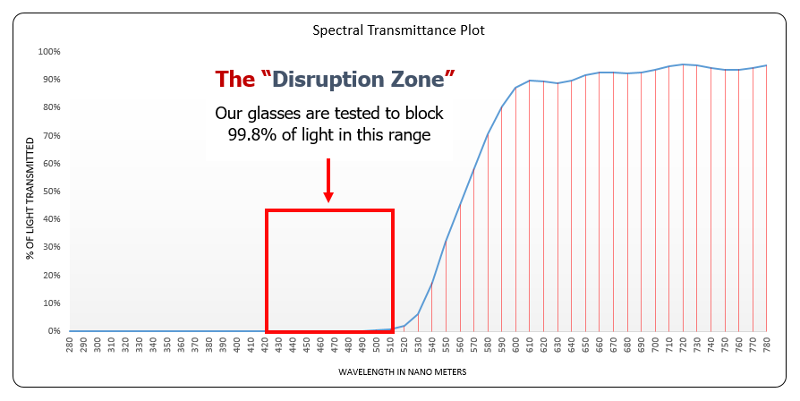
Carbonshades block 99.8% of 400-570 nm, which fully encompasses blue to green, so the largest range of protection according to study 1 (above) and study 2 (below).

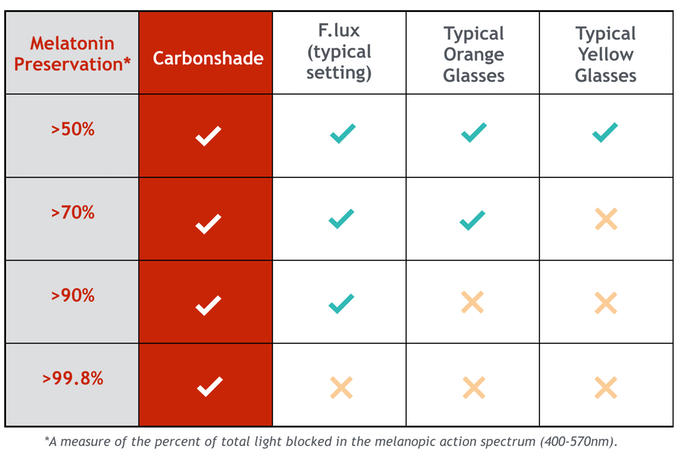
I haven’t seen the spectral transmission data on Carbonshades, although they performed the best on Spectra479’s at-home test.
The popular orange-lensed Skypers block 98% of blue light and probably not too much green (as per the transmission data below and Spectra479’s at-home test).

Affiliate discounts: if you’re still looking for a pair of hot blue blockers, Carbonshade is offering 15% off with the coupon code LAGAKOS and Spectra479 is offering 15% off HERE. If you have no idea what I’m talking about, read this then this.
20% off some delish stocks and broths from Kettle and Fire HERE.
If you want the benefits of ‘shrooms but don’t like eating them, Real Mushrooms makes great extracts. 10% off with coupon code LAGAKOS.
For more steps on how to strengthen your circadian rhythms, the potential importance (and relevance) of blocking BLUE/GREEN, and a discussion of the science… head over to Patreon!
Also many more interesting tidbits and some advice, like who might need to upgrade their blue-blockers.
Three bucks a month for access to all articles and there are many other options. And it’s ad-free and you can cancel if it sucks. Don’t hesitate, there are only a limited number of positions remaining at the $3 level.
Lastly, I’m open to suggestions; please feel free leave a comment or email me directly at drlagakos@gmail.com.
calories proper
Become a Patron!
Save
Save