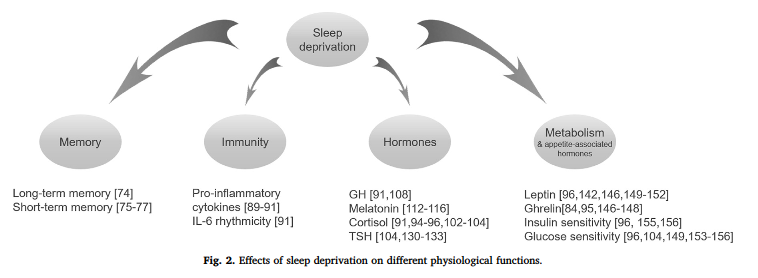
In Nedeltcheva’s notorious weight loss study, those assigned to shorter sleep duration (5.5 hours) lost more muscle and less fat mass than those assigned to adequate sleep duration (8.5 hours). Same diet + same calories + circadian arrhythmia = no bueno.
*I’d say 8.5 hours can be called in this context. I like saying, seasonally, 8-9.5 hours of darkness, most of which should be spent sleeping. Others say 7-9 hours is adequate and <7 is inadequate. I agree with that, too*
Similar outcomes are seen in any number of sleep restriction studies. Figuratively, everything goes down the toilet. But that’s only half of the story.
There are a couple randomized intervention studies on intentional sleep extension and the results are very cool.
The effects of extended bedtimes on sleep duration and food desire in overweight young adults: a home-based intervention (Tasali et al., 2014)
They recruited habitual under-sleepers (<6.5 hours), recorded their sleep behaviors for a week, then gave them individualized behavioral counseling on sleep hygiene with the goal of extending their sleep duration to 8.5 hours. It worked: they were less sleepy, felt more vigorous, and spontaneously ate less sugar and fewer calories. Had the intervention lasted longer than 2 weeks, we would’ve seen body comp improvements as per Nedeltcheva’s study results. IT STARTS WITH SLEEP.
Beneficial impact of sleep extension on fasting insulin sensitivity in adults with habitual sleep restriction (Leproult et al., 2015)
Similar set-up as above: 2 weeks initial monitoring, then 6 weeks where they slept about an hour longer. It worked. Total sleep time positively correlated with indices of insulin sensitivity.
Feasibility and emotional impact of experimentally extending sleep in short-sleeping adolescents (van Dyk et al., 2017)
Kids who normally slept 5-7 hours a night increased that by ~70 minutes for 5 weeks. It worked: reduced symptoms of sleepiness, anger, and confusion! Seriously?
Seriously.
Most of these studies basically just told the participants to go to bed earlier (in evening chronotypes) or sleep a little later (in morning chronotypes). It was individualized and not pharmacological. That means you can do it.
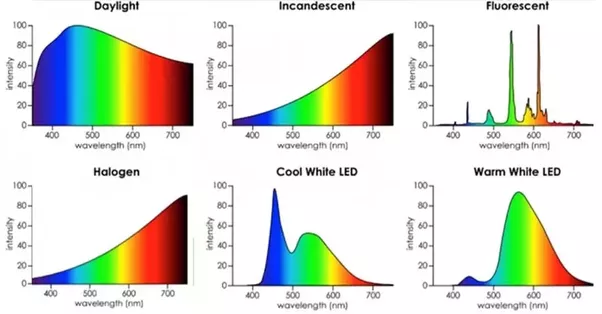
Check it: Blocking nocturnal blue light for insomnia: a randomized control trial (Schecter et al., 2018) CROSSOVER STUDY. Tl;dr: it worked. Blue blockers, FTW.
If you like what I do and want to support it, check out my Patreon campaign!
UPDATED Affiliate links: still looking for a pair of hot blue blockers? Carbonshade is offering 15% off with the coupon code LAGAKOS and Spectra479 is offering 15% off HERE.
If you have no idea what I’m talking about, read this then this.
20% off some delish stocks and broths from Kettle and Fire HERE.
If you want the benefits of ‘shrooms but don’t like eating them, Real Mushrooms makes great extracts. 10% off with coupon code LAGAKOS. I recommend Lion’s Mane for the brain and Reishi for everything else.
Join Earn.com with this link. Get paid to answer questions!
calories proper
Become a Patron!