If you’re healthy, no major complaints, then you probably won’t benefit from tweaking your ‘biome. Ymmv. But if you’re gonna do it anyway, here are some tips (mostly my opinions).
If you’re healthy, no major complaints, then you probably won’t benefit from tweaking your ‘biome. Ymmv. But if you’re gonna do it anyway, here are some tips (mostly my opinions).
Posted in Advanced nutrition, diet, fermentation, fiber, microbiome
Tagged bifidobacteria, diet, fiber, GOS, ketosis, microbiota, nutrition, obesity, Paleo, prebiotics, probiotics
Nuts are good calories.
I’m not a big fan of the omega-6 fatty acid linoleate, but that’s largely in the context of processed foods and confectioneries, where it’s more than likely no longer in it’s native form (Dc9,1218:2n6)… but in the context of unprocessed whole foods (eg, nuts), a little n6 is fine imo.
What are good calories? They’re nutrient-dense and don’t generally lead to overeating… like the opposite of soda and junk food. Nuts are low carb and many are highly ketogenic (eg, Brazils, macadamias, and pecans are ~90%fat). Mr. Ramsey may even approve of macadamias because they have virtually zero PUFAs.
BONUS: magnesium, copper, selenium, many trace minerals and micronutrients, etc., etc.
I’m not saying you should crack open a can of Deluxe Mixed Nuts and sit down with nothing to do other than NOM NOM NOM ALL THE NUTZ. I’m talking about a few nuts with a meal. Possibly earlier in the day (coinciding with LIGHT); nuts are tryptophan-rich and this may improve melatonin onset -> good for circadian rhythms:
In this study, the participants were instructed to eat a serving of almonds (~43g, ~245 kcal) daily for four weeks, at different times of the day (with breakfast, midmorning snack, lunch, or afternoon snack).
Regardless of when the almonds were consumed, the calories were practically completely compensated for. The participants unwittingly ate less other stuff. And in 3 out of 4 of the conditions, the almonds were so satiating that the participants actually ended up eating fewer overall calories.
That, in a nutshell, is what I call “good calories,” and I don’t think it’s too far from Taubes’ original definition… especially because it was accompanied with [modest] reductions in body fat (NS). To be clear, they were instructed to eat more (in the form of almonds), but ended up eating less, BECAUSE ALMONDS. This wasn’t a cross-sectional study, so no healthy user bias or other obvious confounders.
Further, the participants clearly weren’t obesity resistant. They were overweight, obese, or lean with a strong family history of type 2 diabetes. Sam Feltham would’ve been excluded.
This is not an isolated finding: another study showed a dose-dependent response to almonds: 28g or 42g consumed in the morning resulted in a compensatory reduction of hunger and total energy intake at lunch and dinner (Hull et al., 2014). This wouldn’t happen with soda or junk food.
Another study tested ~350 kcal almonds daily for 10 weeks and concluded: “Ten weeks of daily almond consumption did not cause a change in body weight. This was predominantly due to compensation for the energy contained in the almonds through reduced food intake from other sources” (Hollis and Mattes, 2007).
Almonds vs. complex carbs? Almonds, FTW.
1 Brazil nut daily: “After 6 months, improvements in verbal fluency and constructional praxis (two measures of cognitive performance) were significantly greater on the supplemented group when compared with the control group.” ONE FRIGGIN’ NUT!
Walnuts protect against alcohol-induced liver damage (in rats) (Bati et al., 2015) and may improve brain health (in humans) (Poulose et al., 2014).
Pistachios improve metabolic and vascular parameters (Kasliwal et al., 2015).
Meta-analysis (not an intervention study): nut consumption is associated with lower risk of all-cause mortality (Grosso et al., 2015). Yeah yeah yeah, I know, correlation =/= causation. Whatever.
Nuts are good calories. That’s all I’m saying.
Tl;dr: buy these and one of these
, not this
.
For personalized health consulting services: drlagakos@gmail.com
Posted in Advanced nutrition, diet, Dietary fat, empty calories, fat, fiber, insulin, microbiome, microbiota, Protein
Tagged bifidobacteria, body composition, calories proper, diet, empty calories, energy balance, fiber, insulin, nutrition, prebiotics
Fruits and veggies, fermented or otherwise, aren’t the only source of prebiotics in your diet. Eat a whole sardine and some of the ligaments, tendons, bones, and cartilage will surely escape digestion to reach the distal intestine where they will be fermented by the resident microbes.
Salmon skin and the collagen in its flesh, the tendons that hold rib meat to the bone, and maybe even some of the ligaments between chicken bones. All of these are potential prebiotics or “animal fibres.” And it may explain why fermented sausages are such good vessels for probiotics.
“Animal prebiotic” may be a more appropriate term because the food matrix is quite different from that of non-digestible plant polysaccharides. And while I doubt those following carnivorous diets are dining exclusively on steak, these studies suggest it might be particularly important to eat a variety of animal products (as well as greens, nuts, dark chocolate, fermented foods, etc.) in order to optimize gut health.
These studies are about the prebiotics in a cheetah’s diet. Cheetah’s are carnivores, and as such, they dine on rabbits, not rabbit food.
As somewhat of a proof of concept study, Depauw and colleagues tried fermenting a variety of relatively non-digestible animal parts with cheetah fecal microbes (2012). Many of the substrates are things that are likely present in our diet (whether we know it or not).
Cartilage
Collagen (tendons, ligaments, skin, cartilage, bones, etc.)
Glucosamine-chondroitin (cartilage)
Glucosamine (chitin
from shrimp exoskeleton? exo bars made with cricket flour?)
Rabbit bone, hair, and skin (Chicken McNuggets?)
The positive control, fructooligosaccharides (FOS), was clearly the most fermentable substrate; however, glucosamine and chondroitin weren’t too far behind. Chicken cartilage and collagen were also well above the negative control (cellulose). Rabbit skin, hair, and bone weren’t particularly good substrates.
As to fermentation products, collagen, glucosamine, and chondroitin were actually on par with FOS in terms of butyrate production:
Glycosaminoglycans (glucosamine and chondroitin) are found in cartilage and connective tissues (ligaments and tendons) and may have been mediating some of these effects as they’re some of the carbiest parts of animal products. Duck Dodgers wrote about this in a guest post at FTA and in the comments of Norm Robillard’s article (probably elsewhere, too); very interesting stuff.
The authors also mentioned that the different fermentation rates in the first few hours suggests an adaptive component (some took a while to get going), or that certain substrates induced the proliferation of specific microbes. “Animal prebiotics.”
This is particularly noticeable for FOS (solid line), which is a plant fibre that wouldn’t really be present at high levels in a cheetah’s diet, so the microbes necessary to ferment it were probably not very abundant (initially). Chicken cartilage (long dashes), on the other hand, started immediately rapidly fermenting, perhaps because this is more abundant in the cheetah’s diet.
Depauw took this a step further and fed cheetahs either exclusively beef or whole rabbit for a month (2013). Presumably, the beef had much less animal fibre than whole rabbit. When they initially examined fecal short chain fatty acids, there were no major differences between the groups:
However, if you take into consideration that the whole rabbit-fed cheetahs produced over 50% more crap than meat-fed cheetahs, then some other differences become apparent. For example, the concentration of total SCFAs is actually greater in the feces from whole rabbit-fed cheetahs:
edit: la Frite pointed out that the table in the original manuscript is incorrect; the total SCFA numbers are reversed. The excel table above is corrected.
Further, the mere fact that there was 50% more fecal mass per day pretty much confirms way more animal fibre in whole rabbits. And while neither of these studies were accompanied by microbial analysis, a more recent study on cheetahs fed primarily meat, “randomly interspersed with unsupplemented whole rabbits,” showed low levels of Bacteroidetes and Bifidobacteria, two potentially health-promoting groups of microbes (Becker et al., 2014). I suspect this may have been at least partially due to a relative lack of animal fibre, compared to the Depauw’s exclusive whole rabbit diet.
Human digestive physiology and gut microbes are certainly far different from that of a cheetah, but maybe we too receive some prebiotic benefits from these animal fibres… just something to think about next time you’re eating sardines or pork ribs.
Consults are open, contact me if you’re interested: drlagakos@gmail.com
Affiliate links: Still looking for a pair of hot blue blockers? TrueDark is offering 10% off HERE and Spectra479 is offering 15% off HERE. If you have no idea what I’m talking about, read this then this.
Join Binance and get some cryptoassets or download Honeyminer and get some Bitcoins for free!
20% off some delish stocks and broths from Kettle and Fire HERE.
If you want the benefits of ‘shrooms but don’t like eating them, Real Mushrooms makes great extracts. 10% off with coupon code LAGAKOS. I recommend Lion’s Mane for the brain and Reishi for everything else.
Join Earn.com with this link.
Start your OWN Patreon campaign!
Posted in Advanced nutrition, chocolate, fermentation, fiber, gelatin, glycine, microbe, microbiome, microbiota
Tagged bifidobacteria, carbs, chocolate, diet, fiber, GOS, inulin, microbiota, prebiotics, probiotics
From Slate: “Sausage made with bacteria from baby poop isn’t as gross as it sounds.”
and my favorite: “Pooperoni? Baby-poop bacteria help make healthy sausages.”
Much ado about: Nutritionally enhanced fermented sausages as a vehicle for potential probiotic lactobacilli delivery (Rubio et al., 2014)
The media seems to have missed the ball, but not by far. They focused on healthy microbes being incorporated into fermented meats, whereas the scientists seemed to want to make a “healthier” low-salt, low-fat sausage.
The low-salt part seems to partially make sense from a fermentation-perspective: using probiotics instead of salt to reduce the potential for pathogenic microbial contamination. However, I doubt reducing the sodium by 25% will have any appreciable impact on health outcomes. The effect of adding beneficial microbes, on the other hand, might.
They also mentioned making it lower in fat, but that doesn’t make as much sense; I don’t think there’s a big contamination risk of having a higher fat content. #lipophobia
Posted in Advanced nutrition, chocolate, Dietary fat, fat, fermentation, Protein
Tagged bifidobacteria, chocolate, fat, microbiota, nutrition, prebiotics, probiotics, processed food
Bifidobacteria undoubtedly like resistant starch (RS). They bind and hold on tight, an effect mediated by cell surface proteins. Big thanks to Tim Steele for passing along many of the studies cited here. One of said studies showed that treatment of bifidobacteria with proteases abolished the RS binding; but even dead critters would bind if their cell surface proteins were intact (Crittenden et al., 2007).
I suspect fermented foods have this all figured out. The microbes in sauerkraut are going to be embedded in & all around the cabbage polysaccharides; likely protected from digestive enzymes (to a degree) and holding on tight.
Something similar has been shown for galactooligosaccharides (GOS) (Shoaf et al., 2006). In this study, GOS, but not a variety of other fibres, inhibited the binding of pathogenic gut microbes to intestinal epithelial cells.
These mechanisms are likely not mutually exclusive, and both seem like they could benefit the host (us).
Posted in Advanced nutrition, chocolate, diet, Grains, microbiota
Tagged bifidobacteria, calories proper, carbs, diet, fiber, galactooligosaccharides, GOS, insulin, microbiome, microbiota, nutrition, potato, prebiotics, probiotics, resistant starch, starch
For overall health and well-being, fermented foods like sauerkraut and kefir are great. Especially when following a low carbohydrate diet which is generally low in the types of foods which feed the gut microbiome.
For those with gastrointestinal problems, the gut microbiota is probably involved. Whether it is bacterial overgrowth or dysbiosis, gut bugs are usually the culprit. Treatment options vary widely, ranging from global extermination with vinegar & a low fibre diet (as per Jane Plain), or remodeling the microbiome with a prebiotic like galactooligosaccharides. Probiotics like bifidobacteria can help, too, if they’re administered with either prebiotics or fermented foods (they need something to nourish them in transit). Dark chocolate is also an excellent vessel. Resistant starch is another option, although the question remains as to whether or not this is compatible with a low carbohydrate diet.
Resistant starch has been around for a while, and when I was in school it received about 10 minutes of attention during the fibre lecture. But Jimmy Moore and Richard Nikolay have been talking about it a lot lately so I decided to freshen up on the topic. In brief, it can be therapeutic for GI issues, but some studies have shown mixed effects on glucose & insulin metabolism. The former is virtually unarguable, but I found the latter interesting. And the impact of resistant starch on ketosis is included as well.
Posted in Advanced nutrition, clamp, diet, fiber, Grains, insulin, Ketosis
Tagged bifidobacteria, carbohydrates, diet, energy balance, fibre, grains, insulin, ketogenic, ketosis, microbiota, nutrition, prebiotics, protein
Treatment for dietary protein-induced brain fog: dark chocolate with 3% GOS and 10% MCTs. Who’s in?
#IntermediaryMetabolism (bear with me here)
Ketosis from liver’s perspective: increased fatty acid influx & [partial] oxidation causes acetyl-CoA levels to rise dramatically. Concomitantly, gluconeogenesis redirects oxaloacetate (OAA) away from combining with acetyl-CoA via TCA cycle citrate synthesis and toward gluconeogenesis. Since the acetyl-CoA doesn’t have much OAA with which to couple, it does itself to make acetoacetate. Ergo, ketosis, and fortunately liver lacks ketolytic apparatus.
Brain is singing a different tune. Ketones provide ample acetyl-CoA and are efficiently metabolized in the TCA cycle. Ketolysis is not ketogenesis in reverse, else liver would consume ketones.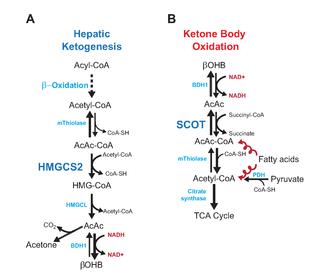
Teleologically speaking (and I don’t really know what that word means), ketones are meant to spare glucose for the brain by replacing glucose as a fuel for peripheral tissues like skeletal muscle and displacing some brain glucose utilization. The former is vital as one of the few sources of “new” glucose is skeletal muscle amino acids, and they would be exhausted in a short amount of time if skeletal muscle kept burning glucose –> incompatible with survival. Getting some of that fuel from fatty acids, ie, ketones, is just way better. Thus, the “glucose sparing effect of fat-derived fuel.” And by “glucose,” I mean “muscle;” and by “fat-derived fuel,” I mean “ketones.” There are numerous intracellular signaling events and biochemical pathways pwned, but that’s the gist of it.
Posted in Advanced nutrition, chocolate, coconut, diet, fiber, liver, microbiota, Protein
Tagged bifidobacteria, chocolate, diet, fiber, GOS, microbiota, prebiotics
Part 1. Guts
Nice review article about the great diversity of carbohydrate-modified diets used in the treatment of gastroin-testiness.
Short-chain carbohydrates and functional gastrointestinal disorders (Shepherd, Lomer, and Gibson 2013)
the full version (click to enlarge, print, and use as a cheat sheet):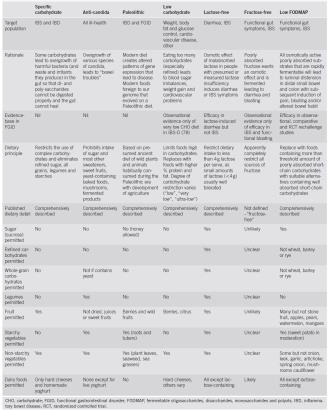
Posted in Advanced nutrition, diet, microbiota
Tagged bifidobacteria, FOS, fructooligosaccharides, galactooligosaccharides, GOS, inulin, microbiota, prebiotics, probiotics
The French Paradox is neither a paradox nor French, really. Red wine isn’t saving the French from a saturated-fat induced heart attack epidemic…. Not to take anything away from red wine, however, as the metabolic effects of red wine (and alcohol in general) are rather interesting.
Background info: alcohol (ethanol) metabolism produces NADH (stick with me here, this article doesn’t get all technical on you I promise).
NADH inhibits gluconeogenesis (Krebs et al., 1969); as such, alcohol lowers blood glucose, regardless of whether if it’s pinot, cabernet, or straight moonshine (Harold R. Murdock, 1971).
Posted in Advanced nutrition, diet, fiber, microbiota, resveratrol
Tagged bifidobacteria, carbs, ethanol, fiber, GOS, inulin, microbiota, mortality, prebiotics, wine
(it’s a “drupe,” not a nut. [Thank you Wikipedia.])
Should almonds be upgraded from “snack” to food? Should almond flour be used in place of some or all white flour? Yes and yes, IMHO.

In 2007, Josse and colleagues did a quick-and-dirty study on almonds and glucose tolerance. They fed a group of volunteers 50 grams of carbs from white bread and either 0, 30, 60, or 90 grams of almonds and then measured blood glucose over the following two hours. “Quick” because they probably had almonds and bread in the refrigerator, and glucometers
in their desk drawers; “dirty” because there were a lot of uncontrolled variables; for example: fiber, protein, and fat content of the test meals differed wildly:
 In a proper study, they might have tried to feed everyone the same amount of fiber, protein, and fat, because each of these is known to affect blood glucose. In any case, the result was pretty cool:
In a proper study, they might have tried to feed everyone the same amount of fiber, protein, and fat, because each of these is known to affect blood glucose. In any case, the result was pretty cool:
Whole almonds dose-dependently blunted the blood glucose response to the test meal. Conclusion: almonds = anti-hyperglycemic. But almonds are complex lil’ things; they’re made of protein, fat, fiber, and a lot of nutrients; so what’s responsible for all the anti-hyperglycemic effect? this post is not simply an academic pursuit; indeed, almond flour
and almond oil
are commercially available, affordable, widely used, and are comprised of different fractions of the almond. So Mori and colleagues decided to study.
Acute and second-meal effects of almond form in impaired glucose tolerant adults: a randomized crossover trial. (Mori et al., 2011)
In this excessively high quality study, the effect of 4 different types of almond preparations on glucose tolerance was assessed.
What was tested (in a FIVE-WAY crossover study):
WA = whole almonds
AB = almond butter
AF = defatted almond flour (remember this stuff? lacks all the bifidogenicity of regular almond flour )
AO = almond oil
V = vehicle: negative control.
Basically, the participants were fed a breakfast of OJ and Cream of Wheat with the equivalent of 33 almonds (42.5 grams) for a total of 75 grams of carbs, and blood glucose was measured over the next 2 hours.
Notable nutritional differences between the almond preparations: they all contain a similar fat content except for the defatted almond flour; whole almonds and almond butter have 2-3 times more fiber than almond flour and almond oil; almond oil has half the protein as all the others.
In brief, no almond preparation affected insulin or free fatty acids.
Whole almonds, almond butter, and almond oil, on the other hand, all blunted the glycemic response. Defatted almond flour, which only really differs in its lack of almond fat, did not. Thus, according to last post, almond fat is a potent bifidogen (i.e., good for gut bacteria); and now we see it’s also responsible for the anti-hyperglycemic effect of almonds. These two effects are probably unrelated, however, as any effect on gut bacteria will take significantly longer than a few hours as the almond fat hasn’t even reached the large intestine by then… (the anti-hyperglycemic effect is evident within 2 hours; the bifidogenic effect noted by Mandalari was 8-24 hours).
OK, almond fat slows the absorption of glucose, so what? this is not exciting… it’s common among most fats- “dietary fat reduces the glycemic index of food.” But this has a greater implication: one could alternatively conclude that almond flour’s lack of fiber was at fault, as dietary fiber is also known to slow glucose absorption. However, almond oil, which has even less fiber than defatted almond flour, was also anti-hyperglycemic. So it’s not the fiber (… perhaps because almond fiber is predominantly insoluble).
With regard to all-things-almonds: almond fat, not almond fiber, is anti-hyperglycemic and bifidogenic (what can’t it do?).
Solution: whole almonds (with meals?), almond oil (with whatever), and regular [non-defatted] almond flour
(for baking?). WRT the latter, get all the benefits, a boost for the gut microbiota, and significantly fewer carbs than with white flour (while actually attenuating the glycemic impact of said white flour).
An argument for almond flour: most baked goods are made with white flour. These foods are predominantly empty calories, the bane of human health and well-being. Substituting almond flour for white flour is one way to decrease the emptiness of those calories, and thus of life itself (it’s gluten-free too).
calories proper
Posted in Advanced nutrition, Dietary fat, empty calories, fiber, gluten, insulin, microbiota, Sugar
Tagged almonds, bifidobacteria, carbs, fat, microbiota, protein