“When we block the D2 receptor in humans, it is expected they will develop glucose intolerance, obesity, and sedentary behavior.” -Jane Plain, in her series on The physiology of body fat regulation. It’s probably true.
Randomized pilot study of cabergoline, a dopamine receptor agonist: effects on body weight and glucose tolerance in obese adults (Gibson et al., 2012)
Cabergoline is primarily used to treat prolinactinoma, or prolactin-secreting tumors. In women (& men apparently), prolactin stimulates milk production; in men, it is associated with the refractory period after orgasm. In both genders, dopamine inhibits prolactin secretion. Cabergoline targets the D2 receptor, but it’s a dirty drug. It’s used off-label for gyno and to improve sexy times (Kruger et al., 2003 <– yes, that was actually tested).
Divide and conquer
16 weeks, 0.5 mg cabergoline twice weekly, obese non-diabetics on a low cal diet. Results: no effect on body weight, but insulin sensitivity increased: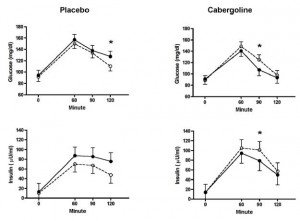
This dosing protocol seems odd, but it worked – prolactin significantly decreased in the treated group. Further, dose timing of dopaminergics (eg, cabergoline, bromocriptine, etc.) seems to be very relevant…
2.5 mg bromo taken twice daily, at 8AM & 8PM, improved circadian GH secretion and modestly reduced leptin levels in obese women (Kok et al., 2006 & 2008).
Cycloset (quick-release bromocriptine) is supposed to be taken immediately upon wakening, which somehow resets the circadian control of hepatic glucose output, ultimately reducing blood glucose levels throughout the day. It is said to inhibit excessive sympathetic tone within the CNS. From Jane’s article & a paper by DeFronzo: hibernation (obesity) = insulin resistance, increased hepatic glucose production, low dopamine: 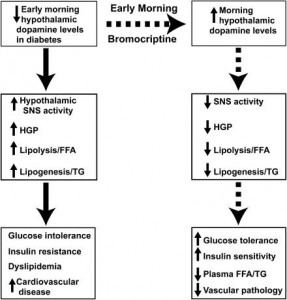
According to DeFronzo’s review, diabetes is characterized by low dopaminergic tone in the early morning which leads to increased central sympathetic activity. Circadian rhythms, prolactin levels, etc., are altered by obesity – timed bromo intake appears to fix this; it makes sense, but I couldn’t find the study which showed the importance of dose timing per se.
Prolactinoma (cabergoline): twice daily 12 hours apart
Metabolic Dysregulation (bromo): once daily in the morning. Lyle McDonald wrote a whole book about bromo (is it really that interesting?).
When it comes to the importance of circadian rhythms, photoperiod, seasonal fluctuations in metabolism, etc., I can get on board. Not a strong, there’s-a-lot-of-hard-evidence-believer, but there are enough tangential studies and anecdotal observations that suggest it’s at least relevant. Probably important.
Another study showed that high leptin levels were associated with high dopamine activity at the D2 receptor, which was reduced in obese subjects (Dunn et al., 2012).
Apomorphine is a another dirty dopaminergic with many useful clinical applications (eg, alcoholism, erectile dysfunction, Alzheimer’s, etc.). Apomorphine inhibits prolactin and stimulates growth hormone, and one study showed that the response to apomorphine was blunted in obesity, further suggesting reduced dopaminergic tone in the obese condition (Brunerova et al., 2013).
“Circulating leptin was also positively associated with stress-induced dopamine release” (Burghardt et al., 2012). However, it’s complicated, modulated differently in different brain regions, and regulated on a long-term basis as leptin therapy corrects many metabolic abnormalities in leptin-deficient humans and some of these, including dopamine signaling, remain corrected even after a month with no leptin injections (Ishibashi et al., 2012). The interplay of leptin and dopamine in the brain is complicated. Leptin reduces hunger and dopamine signaling on the one hand, but on the other hand increased dopamine improves metabolic outcomes. “Location, location, location!” … which, in this case, might refer to location within the brain.
I’ve got a few articles on metreleptin & pramlintide in the tank, mostly animal stuff, but will post them soon.
Affiliate links: It’s 2018, join Binance and get some damn cryptoassets or download Honeyminer and get some Bitcoins for free!
Still looking for a pair of hot blue blockers? Carbonshade and TrueDark are offering 15% off with the coupon code LAGAKOS and Spectra479 is offering 15% off HERE. If you have no idea what I’m talking about, read this then this.
20% off some delish stocks and broths from Kettle and Fire HERE.
If you want the benefits of ‘shrooms but don’t like eating them, Real Mushrooms makes great extracts. 10% off with coupon code LAGAKOS. I recommend Lion’s Mane for the brain and Reishi for everything else.
Start your OWN Patreon campaign!
Join Earn.com with this link and get paid to answer questions online.
