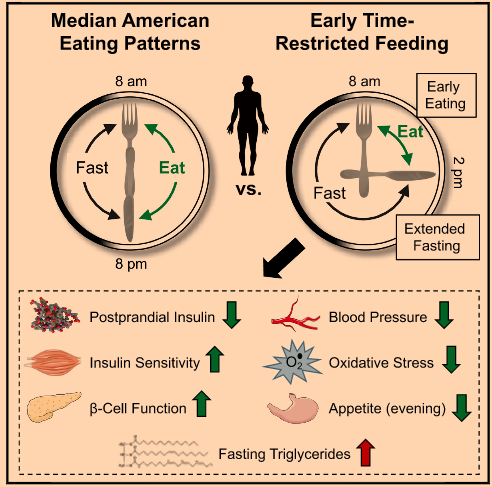
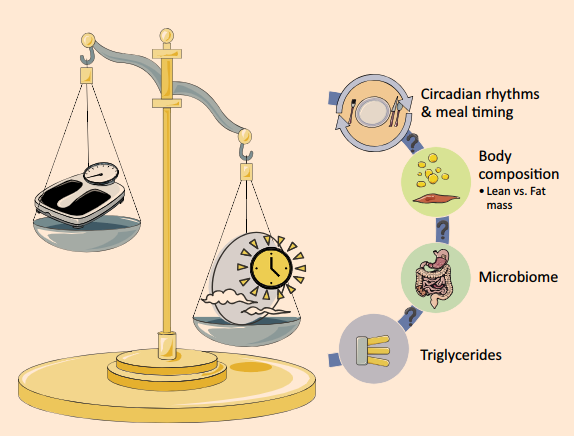
I liked this study for a couple reasons:
Effect of combined use of a low(ish)-carbohydrate, high-protein diet with omega-3 polyunsaturated fatty acid supplementation on glycemic control in newly diagnosed type 2 diabetes: a randomized, double-blind, parallel-controlled trial (Liu et al., 2018)
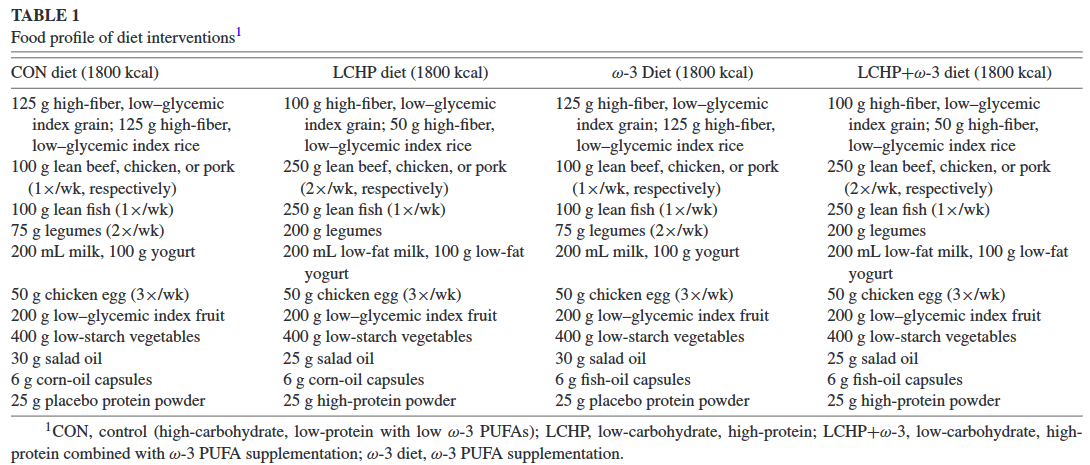
There were FOUR groups: 1) high-carb, low protein (HCLP, aka CONtrol group); 2) LCHP; 3) HCLP+n3; and 4) LCHP+n3. So, instead of just taking a group of people, putting them on LCHP+n3 and comparing the results to baseline, they actually controlled for the variables independently.
All groups were assign 30% fat and the protein was either 17% or 28%. THIS WAS CONFIRMED with serum urea nitrogen and you know how much I like biomarkers! n3 status of the n3 groups were confirmed with plasma n3’s and you know how much I like biomarkers!
Some more details on the study design…
For the rest of this article and more, head over to Patreon! Five bucks a month for full access and there are many other options. It’s ad-free and you can cancel if it sucks 🙂
Consults are open, contact me if you’re interested: drlagakos@gmail.com
Affiliate links: join Binance and get some cryptoassetsor download Honeyminer and get some Bitcoins for free! And now you can mine Bitcoin from your Chromebook!
Still looking for a pair of hot blue blockers? Carbonshade and TrueDark are offering 15% off with the coupon code LAGAKOS and Spectra479 is offering 15% off HERE. If you have no idea what I’m talking about, read this then this.
20% off some delish stocks and broths from Kettle and Fire HERE.
If you want the benefits of ‘shrooms but don’t like eating them, Real Mushrooms makes great extracts. 10% off with coupon code LAGAKOS. I recommend Lion’s Mane for the brain and Reishi for everything else.
Join Earn.com with this link.
Start your OWN Patreon campaign!